Mechanical strength of materials is one of their most fundamental properties, and understanding the effect of material microstructure on its strength was at the origins of physical metallurgy and materials science as scientific and engineering disciplines. The ability to produce stronger steels with higher fracture toughness led to the rise and fall of ancient civilizations. Pure metals are usually strong but ductile, which means that their ultimate strength may be high, but they experience irreversible plastic deformation (yielding) at much lower stresses. About a century ago, Soviet physicist Yakov Frenkel has estimated the shear stress that has to be applied to a crystal in order to cause a relative sliding of its halves past each other, and arrived at the value of about G/10, where G is the shear modulus of material. This estimate is now considered as a theoretical shear strength of material. For iron this estimate results in theoretical shear strength of about 8 GPa, whereas in the reality the shear strength of the best commercially available steels hardly exceeds 1 GPa. The reason for this nearly one order of magnitude discrepancy is the abundance of linear defects in metals – dislocations. The glide of dislocation makes relative sliding of the two crystal halves much easier. Moreover, dislocations are like living creatures – they can interact, multiply, segregate, etc., and their collective behavior is largely responsible for the ductility of metals. How close the two dislocations can get to each other? This depends on their mutual positions and on their Burgers vectors. The dislocations can attract each other and annihilate, provided their Burgers vectors are equal in amplitude and opposite in sign. If they cannot annihilate, they do not like coming too close to each other, because of strong repulsive elastic interaction forces. In metals, except for several special cases such as low-angle grain boundaries, the dislocations rarely come closer to each other than few hundreds of nanometers. So what if we produce a metal sample of submicrometer dimensions? It may happen that it does not contain dislocations at all, and its yield stress will be determined in this case by Frenkel’s formula.
The fact that small metal samples may be ultra-strong has been realized already in the early 1950-s: Conyers Herring has demonstrated that thin whisker of tin can be bent in a fully elastic, reversible manner up to very high deformations unimaginable for bulk tin. You can see a movie of reversible elastic bending of the bi-metallic Au-Fe nanowhisker in our recent publication in ACS Nano. Later, Brenner has carefully tested many metallic whiskers in tension, and demonstrated that their strength increases with decreasing diameter; the thinnest Fe whisker tested by Brenner was about 1 mm in diameter, and achieved an incredible tensile strength of 13 GPa.
In the year 2011, we have published our first work on compression testing of gold nanoparticles fabricated by solid state dewetting method and attached to sapphire wafer. Such particles inherit their orientation from the thin textured Au film they were formed from, which means they are all oriented with their [111] axis normal to the sapphire substrate. The particles are also highly faceted, with their upper flat (111) facet being ideally parallel to the substrate. Thus, it is very convenient to compress them with flat diamond punch. The resulting engineering “stress-strain” curves demonstrated that the particles can be compressed elastically up to very high strains up to 10%. Afterwards, the particles experienced a catastrophic “plastic collapse”, transforming into pancake-like discs. The resulting critical stress (stress at the onset of plastic collapse) exhibited a typical “smaller is stronger” behavior, with the smallest particles achieving the compression strength of 8-9 GPa. For comparison, the yield stress of bulk gold (like the one in the wedding ring) is only 40 MPa! The atomistic computer simulations performed in the same work demonstrated that the plastic collapse of the particles is associated with the nucleation of new dislocations at the upper particle corner. Thus, we have achieved the theoretical strength of gold in our experiments.

Gold particles before and after compression. SEM images (left) and corresponding atomistic model (right).
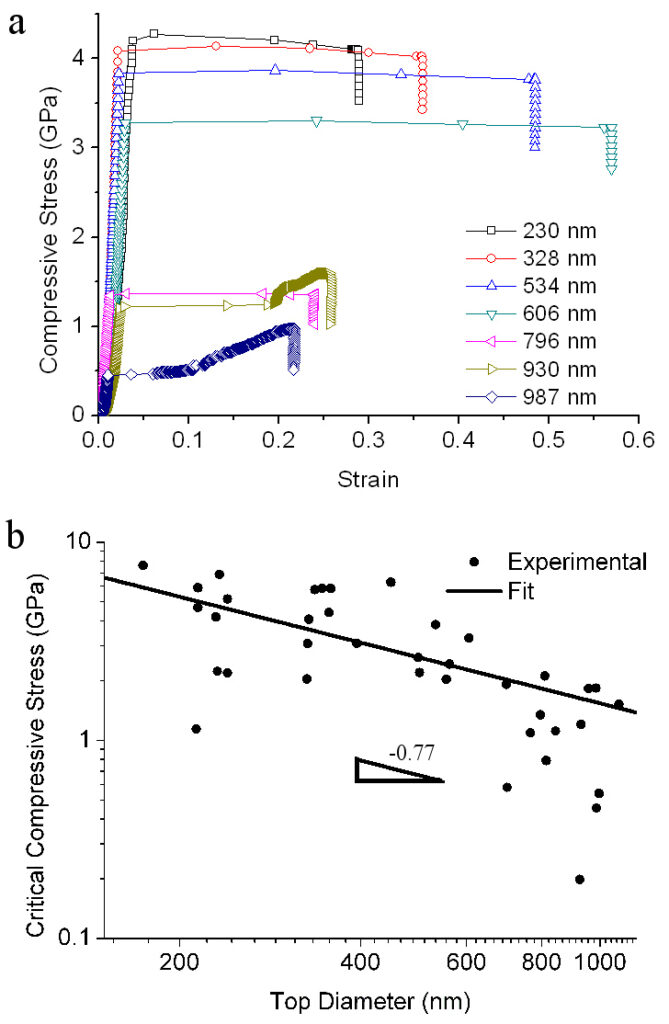
Stress-strain curves of selected Au particles (a) and the dependence of particles strength on the diameter of their top facet.
Later, we performed a similar set of tests on Ni nanoparticles. The smallest of them exhibited an incredible level of strength exceeding 30 GPa. Most interestingly, the strength of some particles tested in the experiments was actually higher than predictions based on atomistic simulations of fully faceted particles. To resolve this contradiction we noted that in the experiment, the facet edges are rounded. The atomistic simulations indicated that “rounding” the sharp facet edges of the particles significantly increases their strength.
You can watch in-situ movie of the nanocompression test of individual Ni nanoparticle here.

Compression tests of Ni nanoparticles. a-b HR-SEM images of particles before (a) and after (b) compression. c-d in situ images of a particle before (c) and after (d) compression. e STEM image of a particle prior to compression. The lattice image in the inset confirms that the particle is single-crystalline. f-g STEM EDS map showing a thin, 3-4 nm oxide layer on the particle surface. The scale bars shown in a-d, e, f and g correspond to 500 nm, 100 nm, 50 nm and 20 nm, respectively.
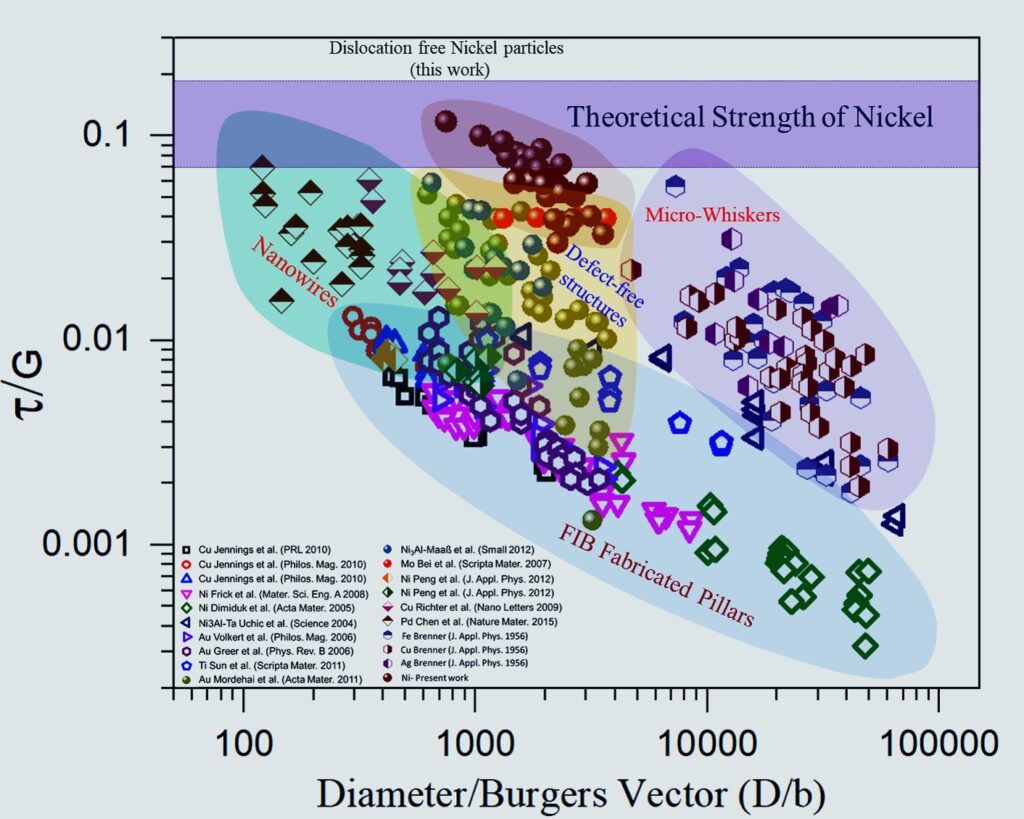
The dependence of normalized resolved shear strength of Ni particles on their normalized diameter, in comparison with the literature data for other types of small-scale samples.
Our recent work on this subject was performed on Mo particles. By manipulating annealing conditions, we were able to produce two different sets of rounded (hemispherical) and highly faceted particles. In both cases, we approached the strength levels of about 50 GPa! This is a geological level of stress, which can be found deep in the Earth interior, and which strongly affects the phase equilibria in the Earth minerals. It is interesting that rounded particles exhibited size-independent strength, while the strength of their faceted counterparts was strongly size-dependent (like in the case of Au and Ni).
Generally, the obtained sequence of compressive strength levels Au<Ni<Mo is consistent with the original model of Frenkel claiming that theoretical strength scales with the material shear modulus.

Record-setting normalized resolved shear strength levels of faceted (red spheres) and rounded (violet spheres) Mo particles, as a function of their projected diameter.
